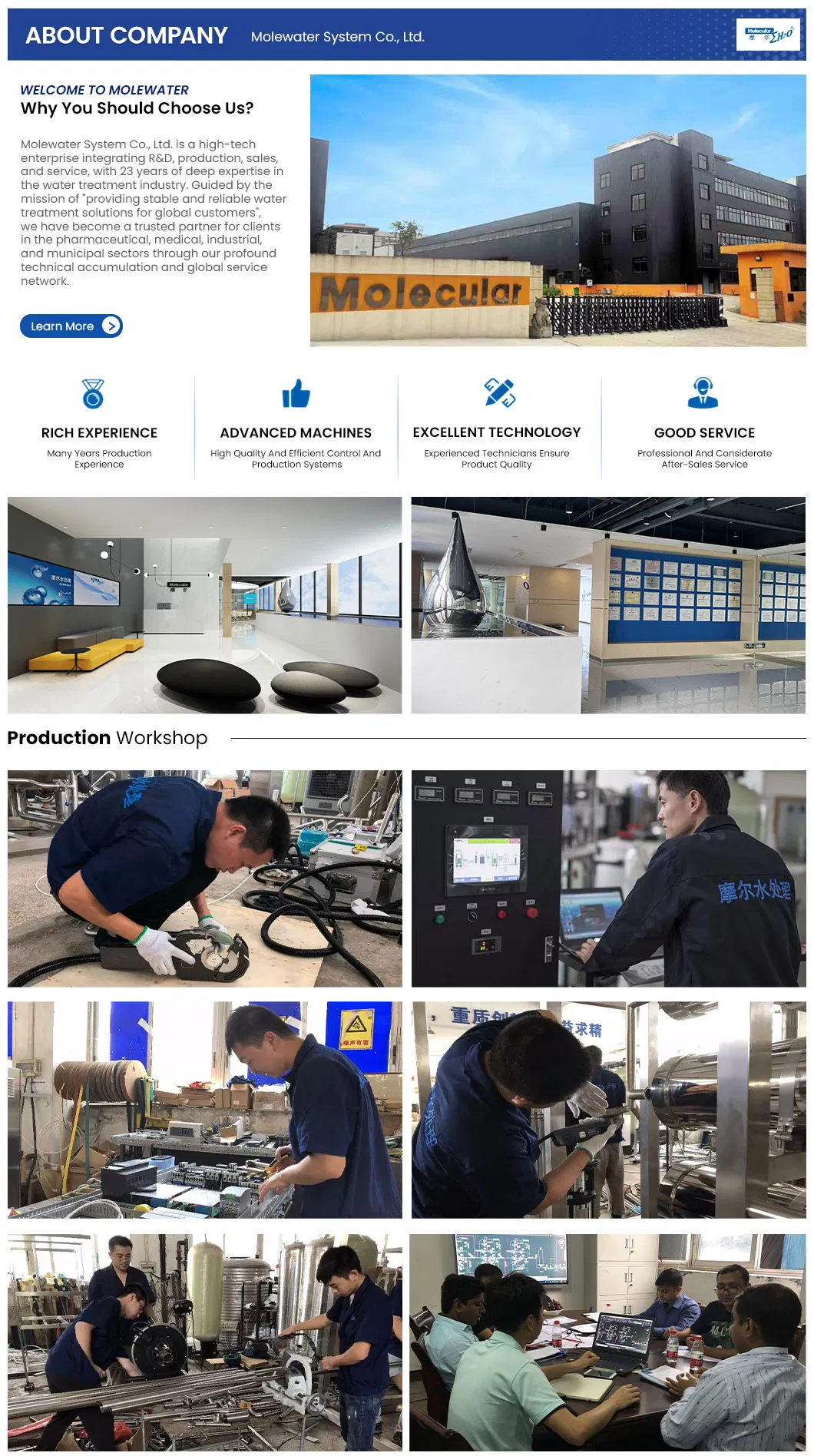
1 / 5











Pharmaceutical purified water system is designed to comply with GMP and USP standards.
| Technical Parameters | |
|---|---|
| Product Name | Pharmaceutical Purified Water Systems |
| Material | SS304 / SS316L |
| System Type | Fully Automated Generation System |
| Purification Technology | Double stage RO+EDI |
| Core Components | Pressure Vessel, High-Pressure Pump, PLC Controller, EDI Module |
| Control System | PLC + HMI (Automatic And Manual Mode) |
| Voltage | 220V / 380V (Customizable) |
| Certification | ISO 9001, ISO 14001, ISO 45001, CE |

Integration of cutting-edge RO and Electrodeionization (EDI) technology ensures ultra-pure water that exceeds pharmacopoeia requirements with a built-in safety margin.

PLC+HMI control system features automatic and manual modes with real-time monitoring and intelligent algorithms to maximize efficiency.

Simplifies installation and maintenance. Components use pharmaceutical-grade stainless steel with a dead-leg-free design to prevent microbial growth.